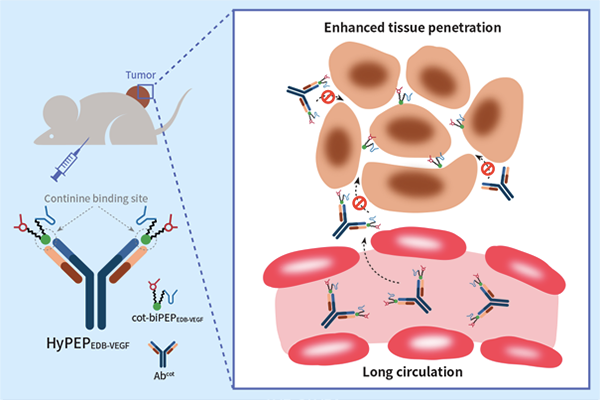
While antibody-based biologics have achieved great success in treating various diseases in the last two decades, the broader use of peptide-based therapeutics has been limited by the short plasma half-life of these agents, reflecting their rapid proteolysis and renal clearance in vivo. Hence, a new molecular platform that can extend plasma half-life and also improve tumor penetration of peptide therapeutics is needed. The limitations of peptide therapeutics have spawned recent efforts to develop alternatives based on the concept of hybrids between peptides and antibodies. As a research project of the Leading Researcher Program funded by National Research Foundation of Korea, Professor Sangyong Jon’s research team has developed a new type of hybrid molecular platform joining a peptide and an antibody; hybrid is designed to overcome a key shortcoming of peptide therapeutics—their short circulation half-life. Unlike previously developed hybrid platforms, the peptide-antibody hybrid developed herein, designated HyPEP-body, was formed through non-covalent interactions between hapten-labeled bispecific peptides and the anti-hapten antibody. A bispecific peptide containing a cotinine tag was synthesized by linking a peptide specific to fibronectin extra domain B (EDB) and a peptide able to bind and inhibit vascular endothelial growth factor (VEGF), yielding cot-biPEPEDB-VEGF. Simple mixing of cot-biPEPEDB-VEGF and anti-cotinine antibody (Abcot) yielded the hybrid complex, HyPEPEDB-VEGF. This complex was able to simultaneously bind to its two target proteins, EDB and VEGF, which resulted in dramatically extended plasma half-life of the HyPEP-body in vivo. Moreover, the HyPEPEDB-VEGF showed significantly increased tumor accumulation and penetration. Finally, it turned out that the HyPEP-body was able to significantly inhibit tumor growth in U87MG human glioma tumor-bearing mice. Taken together, these results suggest that the new peptide-antibody hybrid platform described herein represents a solution to the problem of short duration of action that is characteristic of most peptide therapeutics. On the basis of the aforementioned key points, the new molecular format developed by Professor Jon’s group will also likely attract significant attention from scientists working on targeted cancer therapy using cancer-targeting affinity molecules, including small molecular ligands, peptides, antibodies, and aptamers. This work was published in Angew. Chem. Int. Ed. in January, 2019.
Yu, Byeongjun, Prof. Jon, Sangyong Department of Biological Sciences, KAIST
Homepage: http://www.bionanolab.co.kr
E-mail: syjon@kaist.ac.kr






