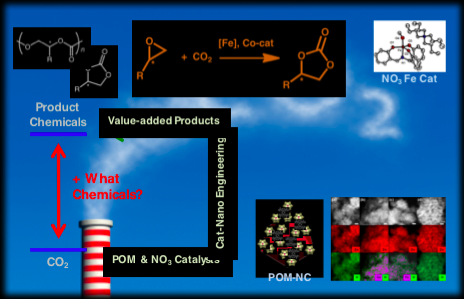
A Saudi Aramco-KAIST CO2 Management Center project (project name: Catalytic Conversion of CO2 toward High Value Commodity Chemicals) led by Professor Sang Woo Han (Department of Chemistry, KAIST) and 6 co-PIs has worked on the development of efficient homogeneous and heterogeneous CO2 conversion catalysts for the production of various high value commodity chemicals.
Because CO2 is abundant, inexpensive, and nontoxic, there have been continuous attempts to develop new chemical processes using CO2 as a renewable C1 resource. Therefore, the development of efficient catalytic processes for the utilization of CO2 will have a significant impact on the chemical industry. Although formic acid and CO are two major products from the CO2 reduction processes, the production of such cheap chemical feedstock cannot be a practical solution due to their weak economic impact on the current industry. Recently, much effort has been devoted to the coupling reaction of epoxide with CO2, as two important products can be made from this reaction, i.e. cyclic carbonate and polycarbonate, which are the main targets of this study. These target chemicals are high-value products that have significant industrial needs. For example, cyclic carbonates with current market size of $1.6 billion have been widely employed in paint-strippers, lithium batteries, biodegradable packaging, and other chemical and medicinal industries. However, given the unfavorable thermodynamics and kinetics for the catalytic CO2 conversion, designing, evaluating, and finding more efficient catalysts for the CO2-insertion chemical transformations still remain a great challenge.
Developing efficient catalysts with a novel approach involving electronic tuning
The development of efficient catalysts for the production of cyclic carbonates and polycarbonates from CO2 is quite challenging and requires cooperative research among various fields of science, such as organic, inorganic, and polymer chemistry as well as nanoscience and computation. For effective cyclic carbonate production, this study has investigated known metal-salen complexes and found their electronic and structural limitations. Thus, with this novel design, the study’s aim is to develop pre-organized catalysts for selective formation of cyclic carbonates from CO2 and various internal epoxides. To this end, a series of (NO3)M complexes (M = Sc, V, Cr, Mn, Fe, Co) utilizing a new NO3 ligand have been recently prepared and a synthetic method to produce a large quantity of precursor (>100 g-scale) for the novel NO3 ligands have been successfully developed. Diverse first-row transition metal complexes supported by an NO3 ligand were synthesized and fully characterized. The coupling reaction of CO2 with propylene oxide was investigated using (NO3)M complexes, which demonstrated that (NO3)M complexes have a high catalytic activity for the propylene carbonate formation.
There are several factors that affect the reactivity and selectivity of catalysts, such as electronic and steric interactions as well as secondary interactions including hydrogen bonding. However, it would be tedious work to repeat catalyst synthesis and activity test without a rational strategy. Thus computational chemistry as a guide for catalyst synthesis was used. Then, based on the known or proposed mechanism of the reaction, the possible catalysts that may enhance the reactivity or selectivity was initially screened by calculating reaction kinetics and thermodynamics.
Developing efficient heterogeneous catalysts for CO2 conversion
In order to achieve high conversion yields and recyclability of cycloaddition reaction of propylene oxide with CO2, this research has focused on the crystallization and immobilization of catalytically active polyoxometalates (POMs). The crystallization of POMs was achieved by the formation of insoluble ionic materials with appropriate counter cations. The complexation of POMs with organometallic cations with appropriate charge, size, shape, and hydrophobicity may lead to the formation of insoluble particles, which is due to the strong ionic interaction between the ionic components with selective catalytic activity. Therefore, POM-based heterogeneous catalysts were prepared by immobilizing different types of transition-metal-substituted Keggin-type POMs on ZnO nanocrystals. These POM-supported ZnO catalysts exhibited efficient, heterogeneous, and reusable catalytic activities for the production of cyclic carbonates from epoxide and CO2.
Han, Sang Woo (Professor, Department of Chemistry)
Homepage: http://ntl.kaist.ac.kr
E-mail: sangwoohan@kaist.ac.kr