The team, working under Professors Seokwoo Jeon and Jihun Oh in the Department of Materials Science and Engineering, has produced a three-dimensional (3D) hierarchical nanostructured catalyst that can effectively reduce the use of gold (Au) by overcoming the limitations of mass transfer during CO2 electrochemical reduction. These results are expected to solve the problem of mass transfer in the field of similar electrochemical reactions and to be applied to a wide range of green energy applications for efficient utilization of electrocatalysts.
With increasing gaseous CO2 emissions and depletion of fossil fuel, research on electrochemical conversion of CO2 into useful chemical compounds has been in the spotlight. The development of a nanostructured catalyst that can improve selectivity to desired chemical compounds and maximize the number of active sites has been intensively studied. However, previously reported complex nanostructures are easily blocked with gaseous CO bubbles during robust electrolysis. The produced CO bubbles hinder mass transfer of the reactants through the electrolyte, causing low conversion rates.
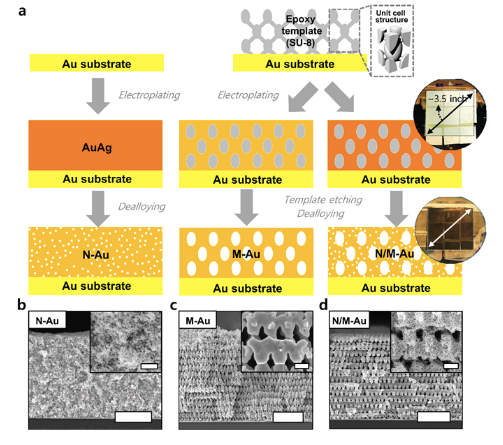
Using proximity-field nanopatterning (PnP), which is effective for fabricating 3D well-ordered nanostructures, and electroplating techniques, the research team successfully fabricated a hierarchically porous gold nanostructure comprising interconnected macroporous channels (200-300 nm) and numerous nanopores (~10 nm). The novel hierarchically porous gold nanostructures show a high conversion rate by inducing efficient mass transfer through the macroporous channels, and high selectivity by producing highly active sites from nanopores, resulting in a high CO selectivity of 85.8 % at a low overpotential of 0.264 V and efficient conversion rate per Au mass that is at maximum 3.96 times higher than that of de-alloyed nanoporous gold electrode.
This work was published online on 4 March 2020 in the journal of the Proceedings of the National Academy of Sciences of the USA (PNAS, Impact Factor: 9.58).

Gayea Hyun, Prof. Jun Tae Song, Prof. Seokwoo Jeon, Prof. Jihun Oh
Dept. of Materials Science and Engineering, KAIST
Homepage: http://fdml.kaist.ac.kr,http://les.kaist.ac.kr
E-mail: jeon39@kaist.ac.kr, jihun.oh@kaist.ac.kr






