Prof. Seung-Hee Lee’s group at KAIST found social isolation during adolescence causes different effects on male and female mice’s behavior and brain maturation. This study revealed that GABAergic inhibitory neurons expressing parvalbumin (PV) in the female left orbitofrontal cortex (OFCL) mature during adolescence They found adolescent social isolation reduced PV expression and social activity in the OFCL, which caused hypersociability in female mice. They genetically deleted the PV gene in the OFCL using the CRISPR/Cas9 system and found it caused an imbalance of excitation and inhibition in the left OFC and caused hypersociability. The study was published on March 1st, 2023 (Adolescent parvalbumin expression in the left orbitofrontal cortex shapes sociability in female mice, Journal of Neuroscience, 43(9) 1555-1571 (2023)).
“We found a sex-specific and lateralized role of the orbitofrontal cortex in shaping social behavior with the maturation of the inhibitory neurons in it,” said Seung-Hee Lee, the corresponding author and a professor in the Department of Biological Sciences and the KAIST Institute for the Biocentury at KAIST.
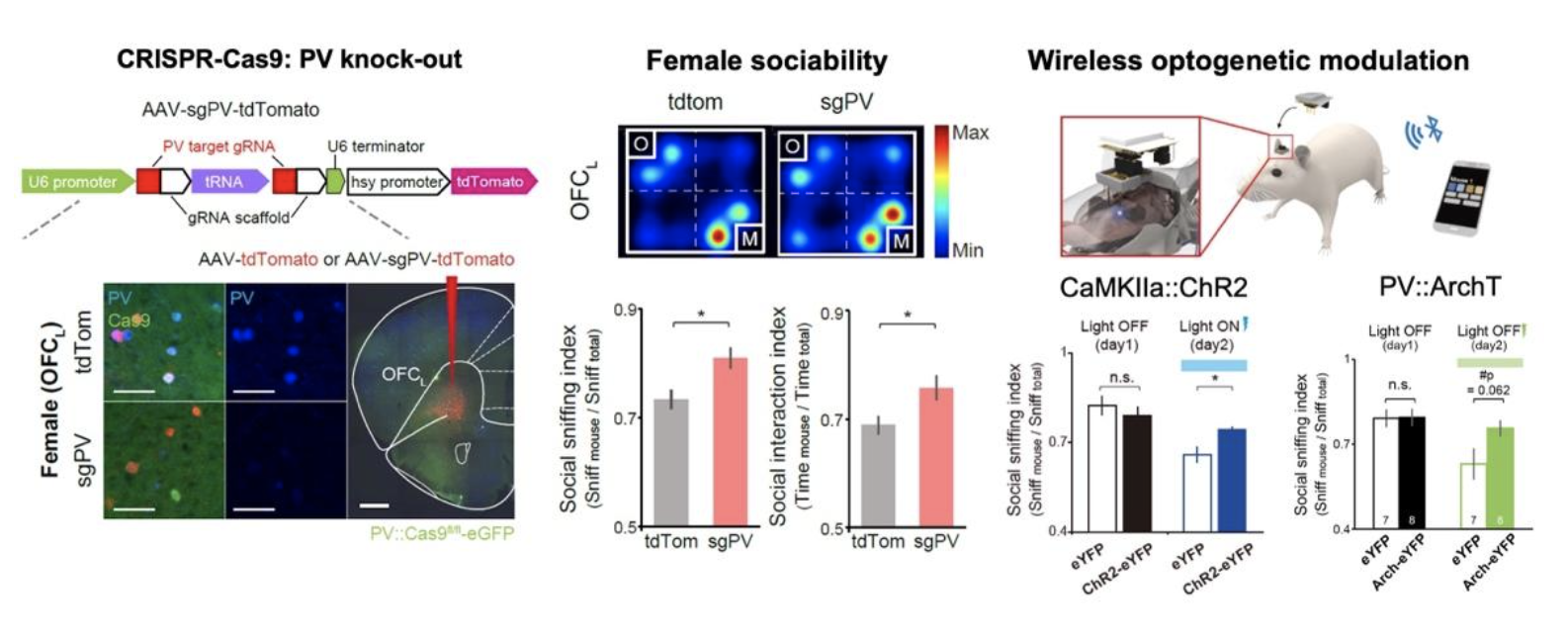
Adolescent social isolation often changes adult social behaviors in mammals. Yet, the sex-specific effects of social isolation and the brain areas and circuits that mediate such changes are still unclear. This study found that adolescent social isolation causes three abnormal phenotypes in female but not male mice: hypersociability, decreased PV+ neurons in the OFCL, and decreased socially evoked activity in the OFCL. Moreover, PV deletion in the OFCL in vivo caused the same phenotypes in female mice by increasing excitation compared with inhibition within the OFCL.
“Previous studies have examined the role of cortical areas in shaping social behaviors mostly in male mice. Our data suggest that adolescent social experience is critical for female mice to shape social behaviors via maturation of PV inhibitory neurons in the OFCL” Prof. Lee said.
“It took us a fair amount of time and effort to examine changes in brain circuits with social isolation in both males and females,” said the first author Yi-Seon Jeon, a graduate student in the Department of Biological Sciences at KAIST. “Once we examined brain samples from both sexes at different ages, it was clear to us that the social experience during adolescence has different meaning in different sexes,” she added. Social experience was critical for PV expression in the OFC. The CRISPR/Cas9-mediated knockdown of PV in the OFCL during late adolescence enhanced sociability and reduced social sniffing-induced activity in adult female mice via decreased excitability of PV+ neurons and reduced synaptic inhibition in the OFCL.
They further proved that balanced inhibition is critical, by optogenetic activation of excitatory neurons or optogenetic inhibition of PV+ neurons in the OFCL. This result demonstrates that the adolescent social experience is critical for the maturation of PV+ inhibitory circuits in the OFCL; this maturation shapes female social behavior via enhancing social representation in the OFCL.
This work was supported, in part, by grants from the National Research Foundation of Korea (NRF-2021R1A2C3012159).
Prof. Seung-Hee Lee, Ms. Yi-Sun Jeon Dept. of Biological Sciences, KAIST
E-mail: shlee1@kaist.ac.kr
Homepage: https://sites.google.com/site/leelab2013/home






