The research teams of Prof. Hye Ryung Byon at KAIST and Prof. Jung Min Joo at PNU developed design strategies for the creation of redox-active organic molecules to be utilized in next-generation non-aqueous redox flow batteries (RFBs). This study was published in ACS Energy Letters on September 1st (ACS Energy Lett. 2021, 6, 3390-3397) and was dedicated as a supplementary cover of the journal.
Energy storage systems have been developed to store the electricity arising from intermittent renewable energy sources reliably. The RFB is one of the most promising storage formats and ensures both a low cost and high safety. However, the volumetric energy density is still poor; it is, 20 times lower for commercial vanadium RFBs than for lithium-ion batteries. Thus, new redox-active materials with high capacities, voltage levels, and a low cost are in strong demand.
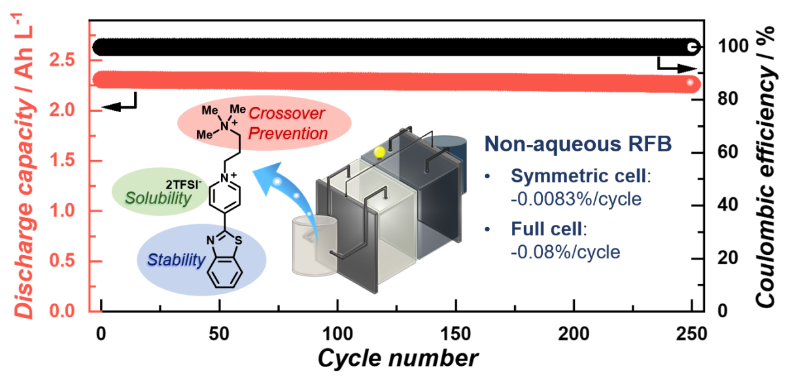
The research teams employed organic molecules as the redox-active materials and investigated strategies to improve their solubility, redox potential, and low crossover between the negative and positive electrode parts. In particular, the teams designed heteroarylpyridinium moieties as negative redox-active molecules. The implemented benzo[d]thiazole group formed an extended -conjugation with the pyridinium core, and the strong resonance reinforced the molecular stability. The introduced 3-(trimethylammonio)propyl cationic group suppressed the crossover phenomenon through the anion-exchange membrane and prevented the molecular interactions from provoking undesired chemical reactions. In addition, the counter anions of bis(trifluoromethanesulfonyl)imide (TFSI) enhanced the solubility of the dicationic pyridinium derivatives in acetonitrile as a solvent. The full RFBs, including this pyridinium-based molecule as the negative electrolyte and ferrocene derivative as the positive electrolyte, showed a capacity- fading rate of 0.08% per cycle for 500 cycles.
Profs. Byon and Joo, who led the joint research, noted that a “A systematically combined study that can synergistically enhance the overall cell performance is highly desirable. We have endeavored to improve all critical parameters, such as the redox potential, solubility, and crossover, on balance.” and ” They also noted that they developed a combined approach that can be simultaneously implemented for non-aqueous RFBs and that will both promote further modification of established redox cores and streamline molecular engineering of new redox-active compounds.”
This work was supported by the Samsung Research Funding & Incubation Center of Samsung Electronics.
Mr. Seongmo Ahn, Prof. Hye Ryung Byon Dept. of Chemistry, KAIST
Homepage: https://www.emdl.kaist.ac.kr
E-mail: hrbyon@kaist.ac.kr






