Graduate School of Medical Science and Engineering (GSMSE)’s professor Pilhan Kim and his team developed a new imaging technique to visualize cellular-level dynamic behaviors in the pulmonary microcirculation in vivo. They revealed that neutrophils could generate dead space in the pulmonary microcirculation by obstructing capillaries and arterioles, resulting in microcirculatory disturbance and acute lung injury (ALI) in sepsis.
Sepsis is a major worldwide health burden, which frequently involves lung injury, called acute respiratory distress syndrome (ARDS; clinical term of ALI). To date, the dynamic status of pulmonary microcirculation in the pathophysiology of ARDS was obscure. Therefore, no effective therapies aimed at pulmonary microcirculation to improve the ventilation-perfusion mismatch, namely “dead space”, in ARDS were available. Unfortunately, cellular level imaging of pulmonary capillaries and flowing cells was technically challenging due to severe tissue motion in the physiologic respiration while lung repeats expansion and collapse.
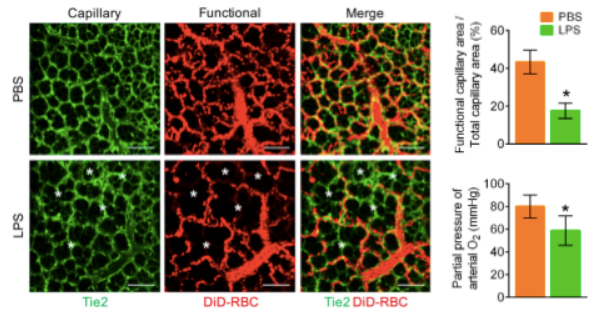
The team utilized a custom-designed video-rate laser scanning confocal and two-photon microscopy system adapted with a newly designed micro-suction-based lung imaging window, which successfully stabilized the lung tissue for real-time cellular-level visualization in vivo. By enabling real-time red blood cell (RBC) flow imaging in the pulmonary microcirculation, the team established a novel quantitative functional parameter, the functional capillary ratio (FCR). Using the new diagnostic approach, they clearly visualized an abnormal perfusion pattern as evidence of decreased FCR in the murine sepsis-induced ALI model (Figure 1).
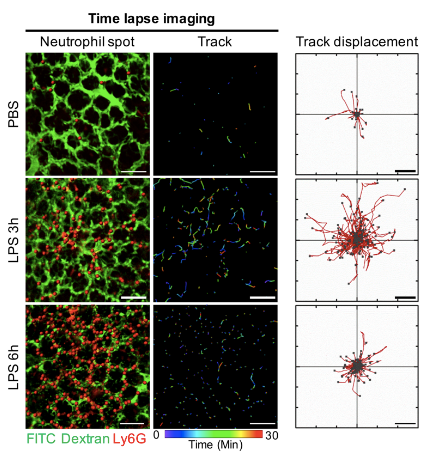
The team identified that neutrophils, actively recruited innate immune cells during systemic inflammation, obstructed the capillaries and interrupted RBCs. With time-lapse imaging of neutrophil dynamics, the team revealed that the motility of intravascular neutrophils initially increased and later decreased, until eventual arrest during the early development of sepsis-induced ALI. Additionally, team successfully captured the entire process of dead space formation in capillaries and arterioles of pulmonary microcirculation by real-time imaging. Furthermore, team has proved that neutrophils produce ROS in situ during capillary entrapment, which aggravates the injury of the vascular endothelium.
Comparing systemic circulating neutrophils and entrapped neutrophils in pulmonary microcirculation, the team discovered that CD11b/CD18 (Mac-1) were highly expressed in the entrapped neutrophils. Using Mac-1 inhibitors including Abciximab, an FDA approved drug for coronary intervention, the team discovered that pulmonary microcirculation was markedly improved and sepsis-induced ALI was ameliorated.
Professor Kim said, “A better understanding of the dynamic role of neutrophils and pulmonary microcirculation during sepsis-induced ALI provides new insight to uncover the pathophysiology and a strategy to develop effective therapeutics for sepsis-induced ARDS.”
This study was published on-line in the European Respiratory Journal (ERJ, IF=12.242) in Jan 2019 (DOI: 10.1183/13993003.00786-2018).
Park, Inwon, Prof. Kim, Pilhan Graduate School of Nanoscience and Technology, KAIST
Homepage: https://ivmvl.kaist.ac.kr
E-mail: pilhan.kim@kaist.ac.kr






