Curvatures of soft matters
Curved surfaces of soft matters define the topological features and significantly influence on their physical properties.
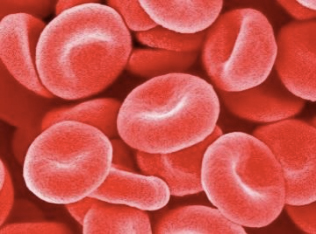
For example, the biconcave disc-like shape of red blood cells that consists of a phospholipid bilayer and an underlying two-dimensional network of spectrin molecules plays a crucial role in the fast gas exchange between hemoglobin and the surrounding medium (Fig. 1).
This shape enables flexible migration of the cells into various vessels including the capillary vessels that are smaller than red blood cells. Another example is found in block copolymers, which are the most popular in the field of nanostructure fabrication of soft matters. These copolymers show a variety of useful curved morphologies such as sphere, cylinder, lamella, and gyroid depending on the composition.
Transformation of curvatures of liquid crystal materials under out of equilibrium condition
Professor Dong Ki Yoon’s group at the Graduate School of Nanoscience and Technology at Korea Advanced Institute of Science and Technology recently reported that a complex nanostructure resembling Udumbara flower, a mysterious flower said to bloom once every 3000 years, can be produced by controlling the curvature of self-assembled structures of liquid crystal (LC) molecules via morphogenesis as shown in Fig. 2. They observed that self-assembled LC structure was reconstructed during the sintering process combined with sublimation and recondensation of LC materials. Their finding was published in Nature Communications this January (check out the following link for the original paper: http://www.nature.com/ncomms/2016/160104/ncomms10236/full/ncomms10236.html).

They utilized smectic LCs, which have layers between bunches of molecules, and added a variety of curvatures to the structure by adjusting different boundary conditions. The term smectic originates from a Latin word smecticus, meaning a sort of soap bubble. The layering structures of smectic LCs closely resemble biological systems such as cell membranes and can therefore provide an interesting insight to morphogenesis in biomaterials.

Sublimation is commonly known as a phase transition from solid to gas phase without passing through the liquid phase while moving from gas to solid is called deposition or condensation. When temperature falls below 0 °C early in mornings in winter, water molecules in gas phase are kinetically deposited onto the substrate like frost on the leaf (Fig. 3). Such sublimation/condensation on the substrate can be considered a sintering process.
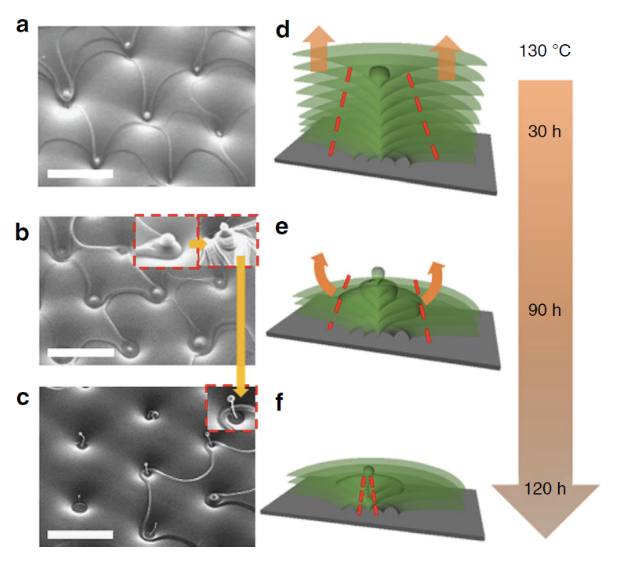
While this kind of morphogenesis on the isotropic material of water is not controllable, the research team utilized smectic LCs in the antagonistic boundary conditions that form the so-called toroidal focal conic domains, i.e., hexagonally arranged holes. When the LCs are kept at 130 °C, a spherical cap forms on the top of the vertical defect line through the rupture, shrinkage, and closure of the uppermost layer of the material (Fig. 4a, d). The uppermost layers around the central cusp region continue to sublimate and shrink with time, leaving remnants of smectic layers around the central line elongated from the cusp; the result is a conical pyramid with a spherical cap at the apex (Fig. 4b, e). The side surface of the pyramid experiences reconstruction. Tangential orientation of the molecules at the LC-air interface associated with the exposed edges of the shrinking layers is replaced by homeotropic orientation, which necessitates hemi-torical rings. As time elapses, LC molecules sublimate from both the areas around the pyramid and from the conical side surface of the pyramid, forming a stalk of the Udumbara flower with a spherical cap at the top and hemi-torical rings shrinking into small beads beneath it (Fig. 4c, f).
This work illustrates that controlling self-assembling behaviors of soft matters under the non-equilibrium conditions could open a way to create such small-feature structures as well as more nontrivial morphologies.