The formation of organs in a living organism is triggered by an agglomeration of primitive cells that mature into three-dimensional structures through morphogenetic processes. To date, a wide range of biomaterials have been attempted to prompt the total or partial replacement of damaged organs and/or tissue structures. Particularly, it is important to properly shape the biomaterials to use as scaffolds. Methodologies such as mold casting and electrospinning had been suggested. However, it has been challenging to achieve exquisite control over cell assembly to produce organ-like structures.
Researchers at Korea Advanced Institute of Science and Technology, led by Professor Sung Gap Im in the Department of Chemical and Biomolecular Engineering, tackled this problem by applying a new vapor-based polymer coating technique, initiated chemical vapor deposition (iCVD) process, to a paper substrate. Based on this technique, they developed a method to bind cell-encapsulated hydrogel on the surface of the iCVD-modified paper substrate and produced 3D cell culture scaffolds using simple paper origami as can be seen in Fig. 1. Their findings were reported in PNAS last December (http://www.pnas.org/content/112/50/15426.abstract).
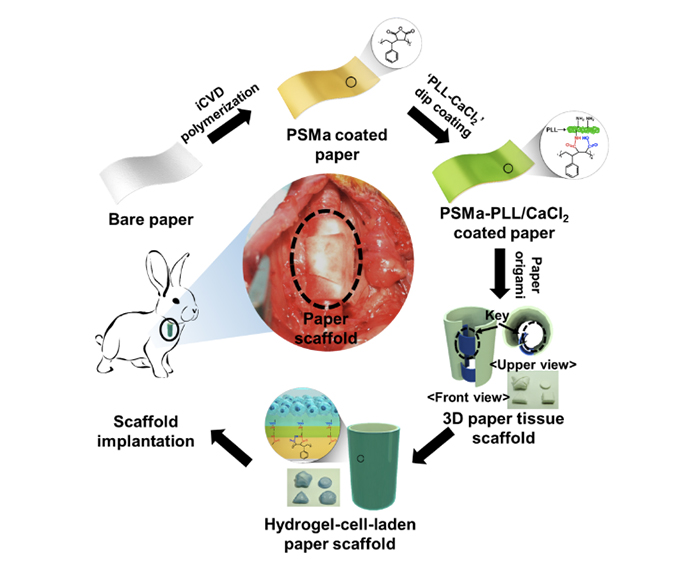
Initiated chemical vapor deposition to functionalize paper surface
Paper is an interesting substrate for biotechnology as it is naturally occurring, biocompatible, non-toxic, and cost-effective material capable of making desired shapes by origami. Also a myriad of paper types are readily available with different mechanical properties and porosity, allowing one to choose an appropriate paper matching with the desired tissue to be regenerated. However, paper becomes extremely weak when wet, which is not suitable for implantation. In addition, the surface of a paper needs to be modified to attract target cells.
The researchers solved these problems by using iCVD process, which enables coating the paper surface conformally with functional polymeric thin films. The process involves flowing of a vaporized monomer and a radical initiator into the iCVD chamber followed by polymerization of adsorbed monomers on the substrate surface via free radical polymerization mechanism.
They demonstrated that iCVD is an advantageous way to chemically modify the surface of the paper to build mechanical strength and introduce desired surface functionality while keeping its macroporous structure, biocompatibility, and foldability. Poly(styrene-co-maleic anhydride) was coated on paper via iCVD process and reacted with poly-L-lysine (PLL) containing Ca2+ions. This occurred when the modified paper was dipped in the PLL solution so that the paper could be coated with amine groups. At this stage, this bio-functionally modified paper could be folded freely into complex 3D tissue-like structure by an origami-based method. Computer aided design (CAD) was used to find out planar figures that could be folded into the 3D target shapes without using a toxic adhesive. Then cell-loaded alginate solution was applied to form alginate hydrogel, which was attached firmly to the surface-modified paper substrate based on the electrostatic interaction mediated by the divalent Ca2+ions.
Successful trachea tissue regeneration
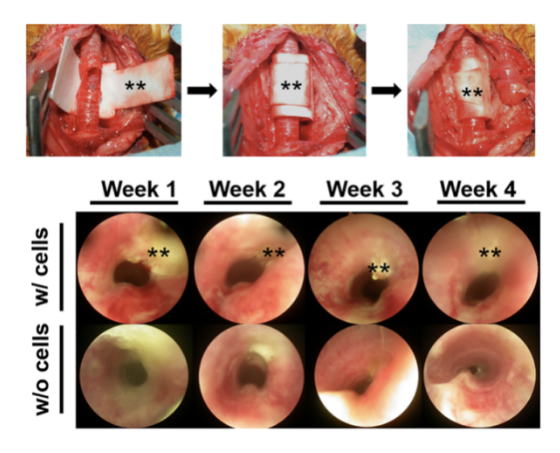
In collaboration with Professor Nathaniel S. Hwang at Seoul National University and Professor SeongKeun Kwon at Seoul National University Hospital, the research showed a great potential for the hydrogel-laden paper scaffolds for implantation. After confirming biocompatibility and permeability with nutrients and oxygen, they fabricated a cylindrical paper scaffold containing rabbit articular chondrocytes in the hydrogel and placed it in a rabbit with a severely damaged trachea. Trachea tissue regeneration was clearly observed after culturing in vivo for 6 weeks without any tissue granulation and signs of immune response (Fig. 2). Professor Kwon stated, “Compared to synthetic trachea scaffolds reported previously, the developed cell-laden paper scaffold system shows an excellent biocompatibility.” He also added that it was remarkable to see no sign of degradation in the mechanical properties of paper in in vivo environment, indicating the advantage of this method.
This research was supported in part by the Ministry of Health and Welfare of Korea (Grants HI13C1789010014, HI13C0451020013, and HI14C15410100); National Research Foundation of Korea funded by the Ministry of Education, Science and Technology (Grant NRF-2012M3A9C6050102); and by the Advanced Biomass R&D Center of the Global Frontier Project funded by the Ministry of Science, Information & Communication Technology (ICT), and Future Planning (ABC-2011-0031350).